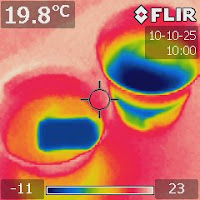
 |
| Figure 1. An IR image of a freshwater cup and a saltwater cup after an ice cube was added to each. |
However, the science behind these questions are not that easy. To explain the results, we will probably need some reasoning at the molecular level, which is not at all easy for lower-grade students. But that is what we hope them to learn. These explorations require not only hands-on but also minds-on, which is why they are so great.
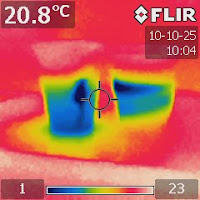 |
| Figure 2. An IR image take after four minutes showing the convection in the freshwater cup. |
Had they had an IR camera, the thermodynamic processes would have been much more obvious. Figures 1-4 show a series of IR images taken to reveal what happened in the two cups after an ice cube was added.
The IR images show that ice molt faster in freshwater because cold molten water can sink to the bottom and warmer water at the bottom is pushed to rise. This process, called convection, runs continuously to carry heat from the whole cup to melt the ice cube.
In the case of saltwater, the cold water just sat at the top. The only explanation of this is that saltwater is denser so molten freshwater from the ice cube cannot sink, even if it is colder. Somehow, saltwater provides greater buoyancy that counters the thermal buoyancy.
Figure 4 shows that sixteen minutes later, the cold front still had not reached the bottom. This means that not only convection slowed down but also conduction was very slow.
Recall our finding that a cup of saturated saltwater can spontaneously develop a temperature gradient from bottom up. This experiment provides a direct evidence that supports the theory that the temperature gradient can be created by the salinity. However, this evidence is not decisive, as the phenomenon reported here happens in an unsaturated solution whereas the small temperature gradient only exists in a saturated solution.
The puzzle still remains unsolved.
PS: Sprinkling some salt to an ice cube seems to accelerate the melting process. This seems to be in contradiction with the observation that ice melts more slowly in saltwater. This is where a lot of people are confused. The physics behind the two processes is different, even though they involve exactly the same chemical ingredients--just water in two different phases and salt.
The IR images show that ice molt faster in freshwater because cold molten water can sink to the bottom and warmer water at the bottom is pushed to rise. This process, called convection, runs continuously to carry heat from the whole cup to melt the ice cube.
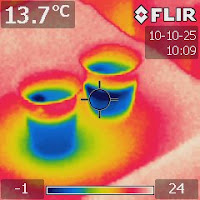 |
| Figure 3. An IR image taken after nine minutes showing the cooling effect at the bottom as indicated by the greenish halo. |
Figure 4 shows that sixteen minutes later, the cold front still had not reached the bottom. This means that not only convection slowed down but also conduction was very slow.
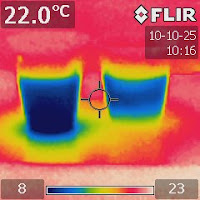 |
| Figure 4. 16 minutes later... |
The puzzle still remains unsolved.
PS: Sprinkling some salt to an ice cube seems to accelerate the melting process. This seems to be in contradiction with the observation that ice melts more slowly in saltwater. This is where a lot of people are confused. The physics behind the two processes is different, even though they involve exactly the same chemical ingredients--just water in two different phases and salt.











